Polycystic ovary syndrome (PCOS) is the most common endocrinopathy in women of reproductive age. A leading cause of infertility and pregnancy complications.
Associated with insulin resistance, metabolic syndrome, non-alcoholic fatty liver disease, and increased risk of developing type 2 diabetes.
Main treatment goals are to reduce hyper-androgenism or to induce fertility.
Letrozole and clomifene are considered first-line pharmacological treatments for infertility in PCOS. Increasing evidence suggests that letrozole may be superior to clomifene.
Definition
Polycystic ovary syndrome (PCOS) includes symptoms of hyper-androgenism, presence of hyper-androgenaemia, oligo-/anovulation, and polycystic ovarian morphology on ultrasound.
Incidence
PCOS affects 6% to 8% of women of reproductive age in the US using the 1990 National Institutes of Health criteria, 7% to 13% of women using the Androgen Excess and PCOS Society criteria, and 8% to 13% of women using the 2003 Rotterdam criteria.[7][8][9] The prevalence does not appear to vary across different regions of the world, as prevalence studies in Greece, Spain, and the UK reveal similar estimates.
A questionnaire-based study suggested that Mexican-Americans, a group at high risk for insulin resistance and the metabolic syndrome, may have a higher prevalence of PCOS.[13] One small preliminary study suggested a high rate of PCOS in indigenous Australians.[14] There have been no prospective studies that document incidence rates for PCOS.
PCOS accounts for 80% to 90% of cases of hyper-androgenism in women. In one large series of women presenting with androgen excess or ovulatory dysfunction, approximately 80% had PCOS, 3% had the hyper-androgenism-insulin resistance-acanthosis nigricans syndrome, 1.5% had 21-hydroxylase-deficient non-classic adrenal hyperplasia, 0.6% had 21-hydroxylase-deficient classic adrenal hyperplasia, and 0.2% had androgen-secreting tumours.
Men in families with PCOS may have manifestations including excessive hairiness, premature male-pattern baldness, elevated levels of dehydroepiandrosterone sulfate, abnormal hormonal responses to dynamic testing, and aberrations in insulin sensitivity and secretion.
Aetiology (Causes)
The aetiology of PCOS is unknown. It is a syndrome wherein multiple systems are affected and the site of the primary defect is unclear. Various lines of evidence have supported primary defects in the hypothalamic-pituitary axis, postulating increased amplitude and frequency of pulses of luteinising hormone, or defects involving the ovaries through an intrinsic problem leading to androgen over-production.
Some theories postulate defects in insulin sensitivity with insulin resistance leading to compensatory hyper-insulinaemia.
PCOS appears to be inherited as a common complex disorder. Multiple genes, each with mild or moderate effects on overall disease risk, are likely to be involved. One twin study calculated the heritability of PCOS at 70%, suggesting that most PCOS risk depends on genetic factors.
The actual PCOS susceptibility genes have yet to be definitively identified. A large genome-wide association study conducted in Chinese individuals robustly identified genetic variants in or near 3 genes (DENND1A, THADA, LHCGR) as risk factors for PCOS.[20] Variation in DENND1A was subsequently associated with PCOS in several European-origin cohorts.
A subsequent genome-wide association study in Chinese individuals increased the number of susceptibility loci to 11.[25] Three genome-wide association studies in European-origin cohorts increased the total number of PCOS susceptibility loci to 19.
Most loci discovered in one ethnic group appear to affect PCOS risk in other ethnic groups, suggesting an ancient origin of PCOS.
Susceptibility loci include genes for the luteinising hormone receptor, the follicle-stimulating hormone receptor, and the follicle-stimulating hormone beta sub-unit, suggesting a key role for disordered gonadotrophin function in PCOS.
Epigenetic changes (DNA alterations independent of the primary nucleotide sequence; e.g., DNA methylation) may also play a role in PCOS susceptibility, as evidenced by differential X-chromosome inactivation in PCOS.
↚
Pathophysiology of Polycystic ovary syndrome
The pathophysiology of PCOS is not well understood, mainly due to lack of knowledge of the location of the primary defect. There are several candidates: ovary, adrenal, hypothalamus, pituitary, or insulin-sensitive tissues. It is possible that there are subsets of women with PCOS wherein each of these proposed mechanisms serves as the primary defect.
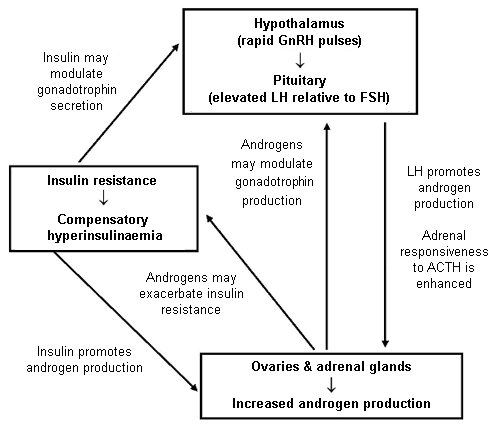 |
| pathogenic factors in PCOS |
Investigations have elucidated some of the interactions between these systems. Insulin resistance leads to compensatory insulin hypersecretion by the pancreas in order to maintain normoglycaemia. The resulting hyperinsulinaemia promotes ovarian androgen output and may also promote adrenal androgen output.
High insulin levels also suppress hepatic production of sex hormone-binding globulin, which exacerbates hyper-androgenaemia by increasing the proportion of free circulating androgens. Another factor that promotes ovarian androgen output is the fact that women with PCOS are exposed long term to high levels of luteinising hormone.
This luteinising hormone excess seems to be a result of an increased frequency of gonadotrophin-releasing hormone pulses from the hypothalamus. The abnormal hormonal milieu may also contribute to incomplete follicular development that results in polycystic ovarian morphology.
Classification (Degrees)
There is no formal classification system for PCOS. The following has been proposed:
- Mild PCOS (accounts for 16% of affected women): characterised by irregular periods, polycystic ovaries on ultrasound, mildly elevated androgen concentrations, normal insulin concentrations, unknown long-term risks
- Ovulatory PCOS (accounts for 16% of affected women): characterised by normal periods, polycystic ovaries on ultrasound, elevated androgen concentrations, increased insulin concentrations, unknown long-term risks
- Hyper-androgenism and chronic anovulation (accounts for 7% of affected women): characterised by irregular periods, normal ovaries on ultrasound, elevated androgen concentrations, increased insulin concentrations, potential long-term risks
- Severe PCOS (accounts for 61% of affected women): characterised by irregular periods, polycystic ovaries on ultrasound, elevated androgen concentrations, increased insulin concentrations, potential long-term risks.
↚
How to reach a diagnosis in a case of PCOS?
There are no pathognomic features that suggest PCOS, and it is largely a diagnosis of exclusion.
History
Common features include hirsutism (affecting 60%), acne (20%), and scalp hair loss (5%); irregular and infrequent periods (often <8 per year); weight gain; and infertility.
Symptoms typically begin at the time of puberty and, in younger women, may be difficult to distinguish from the irregular menses that often occur in the first year after menarche.
Up to 85% of menses are anovulatory during the first year after menarche, and up to 60% in the third year; increased body mass index is a predictor of persistent anovulation. It has been suggested that all 3 Rotterdam criteria or at least both oligo-ovulation and hyper-androgenism must be present to diagnose PCOS in a teenager.
- Infrequent/reduced menstrual bleeding or amenorrhoea should be present for at least 2 years after menarche (or primary amenorrhoea at age 16).Oligo- or anovulation may manifest as menstrual bleeding at intervals of less than 21 days or greater than 35 days.
- Polycystic ovarian morphology should include increased ovarian size (>10 cm³)
- Biochemical hyper-androgenaemia should be present.
Physical examination
- Hirsutism is the most characteristic physical examination finding. Acne and/or alopecia may be present. The degree of hyper-androgenism is typically mild to moderate. Frank virilisation is rare.
- Women may have elevated blood pressure as a part of the hypertension sometimes associated with this syndrome.
- Acanthosis nigricans, usually subtle, may be seen more in obese women with PCOS.
 |
Acanthosis nigricans involving the axilla of an obese white womanFrom the collection of Melvin Chiu, MD, UCLA; used with permission |
- Sweating or oily skin may occur.
Investigations
- The diagnosis of PCOS can be made when infrequent/reduced menstrual bleeding also exists.
- If infrequent/reduced menstrual bleeding is not present in a hirsute woman, evaluation for presence or absence of ovulation (by luteal-phase progesterone measurement or basal body temperature monitoring) should be performed. Anovulatory cycling may occur, particularly in hirsute women. If such measures are consistent with anovulation, PCOS may be diagnosed in hirsute women.
- If the hirsute woman is found to be ovulatory, the next step is to perform a transvaginal ultrasound to examine the ovaries. If polycystic ovarian morphology is documented, then PCOS can be diagnosed.
- If hirsutism is not present, serum androgens should be measured to evaluate for hyper-androgenism.The most commonly measured androgens are total and free testosterone and dehydroepiandrosterone sulfate. Biochemical hyper-androgenism is best assessed by calculated free testosterone, free androgen index, calculated bioavailable testosterone, or by high quality testosterone assays such as liquid chromatography mass spectrometry or extraction/chromatography affinity immunoassays. If any of these are elevated, the diagnostic sequence is the same as when hirsutism is present.
- If hirsutism is not present, all androgen levels are normal, and there is a history of infrequent/reduced menstrual bleeding, an ovarian ultrasound should be performed. Combined with such a history, polycystic ovarian morphology allows a diagnosis of PCOS to be made (Rotterdam criteria only).
- Levels of androstenedione may be checked if other androgens are normal. This may increase the number of women identified as hyper-androgenaemic by 10%.
- Checking dehydroepiandrosterone is of little value.
- Thyroid-stimulating hormone, prolactin, and 17-hydroxyprogesterone should be measured in all women to exclude disorders that may resemble PCOS (thyroid dysfunction, hyperprolactinaemia, and 21-hydroxylase-deficient adrenal hyperplasia, respectively).
- In uncertain cases, elevated luteinising hormone/follicle-stimulating hormone ratio may support a diagnosis of PCOS. However, it is not diagnostic.
- Given the high frequency of insulin resistance and metabolic syndrome in PCOS, an oral glucose tolerance test and fasting lipid panel should be performed in all women with the diagnosis, particularly those with obesity, to evaluate metabolic risk factors.
- Emerging research suggests that pelvic ultrasound may be replaced by measurement of circulating levels of anti-Mullerian hormone; however, this has not yet been recommended for general practice.
Defferential diagnosis
- 21-hydroxylase deficiency
- Thyroid dysfunction
- Hyperprolactinaemia
- Cushing's syndrome
- Androgen-secreting neoplasms
- Syndromes of severe insulin resistance
- Androgenic/anabolic drugs
- Hypogonadotrophic hypogonadism
- Premature ovarian failure
- Apparent cortisone reductase deficiency
Diagnostic criteria
- Hyper-androgenism and/or hyper-androgenaemia
- Oligo- or anovulation
- Exclusion of other disorders.
- Hyper-androgenism and/or hyper-androgenaemia
- Oligo- or anovulation
- Polycystic ovaries on ultrasound.
 |
| Polycystic ovarian ultrasoundFrom the collection of Dr M.O. Goodarzi; used with permission |
- Hyper-androgenism (hirsutism) and/or hyper-androgenaemia
- Ovarian dysfunction: oligo-anovulation and/or polycystic ovarian morphology
- Exclusion of other disorders.
